Abstract
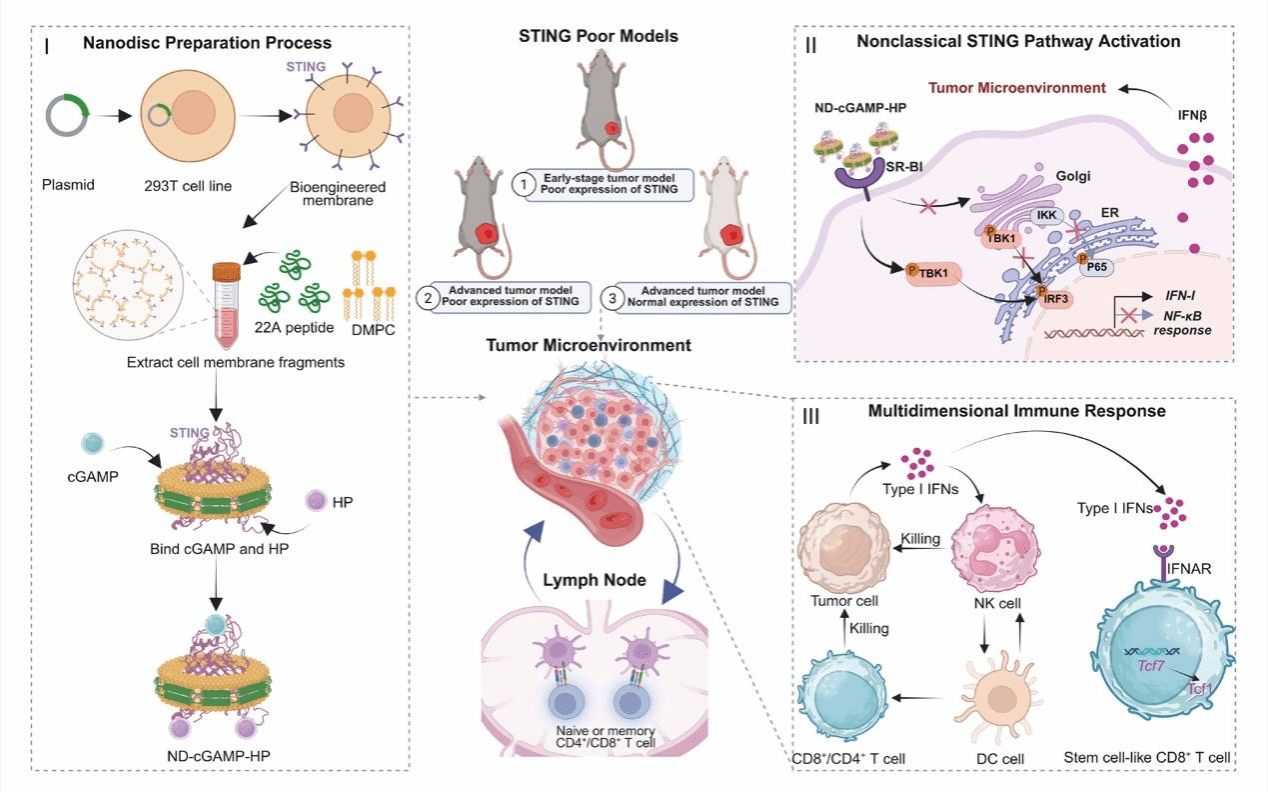
The epigenetic silencing or remarkably diminished expression of STING in cancer cells, along with the structural and functional impairment of the endoplasmic reticulum (ER) and Golgi apparatus, represents a unique mechanism of tumor immune escape and poses an important challenge for STING-targeted therapies. Here, we develop a cell membrane-derived nanodisc system (ND-cGAMP-HP; HP, heparin), which is capable of presenting activated STING proteins in their native form by means of cell membrane-directed display and biological self-assembly techniques. It can directly introduce activated STING protein to tumor cells and circumvent the translocation process between the ER and Golgi apparatus, selectively activating the IFN-I signaling pathway without initiating the inflammation-related pathway NF-κB. ND-cGAMP-HP triggers potent cellular immune responses and remodels the tumor immune microenvironment. Moreover, it augments immune memory by promoting the differentiation of TCF1+ stem cell-like T cells. We thus manifest a strategy based on STING therapy that does not depend on the ER and Golgi apparatus pathways to activate the IFN-I pathway, for cancer immunotherapy.
Link:https://www.nature.com/articles/s41467-026-71363-6