Abstract
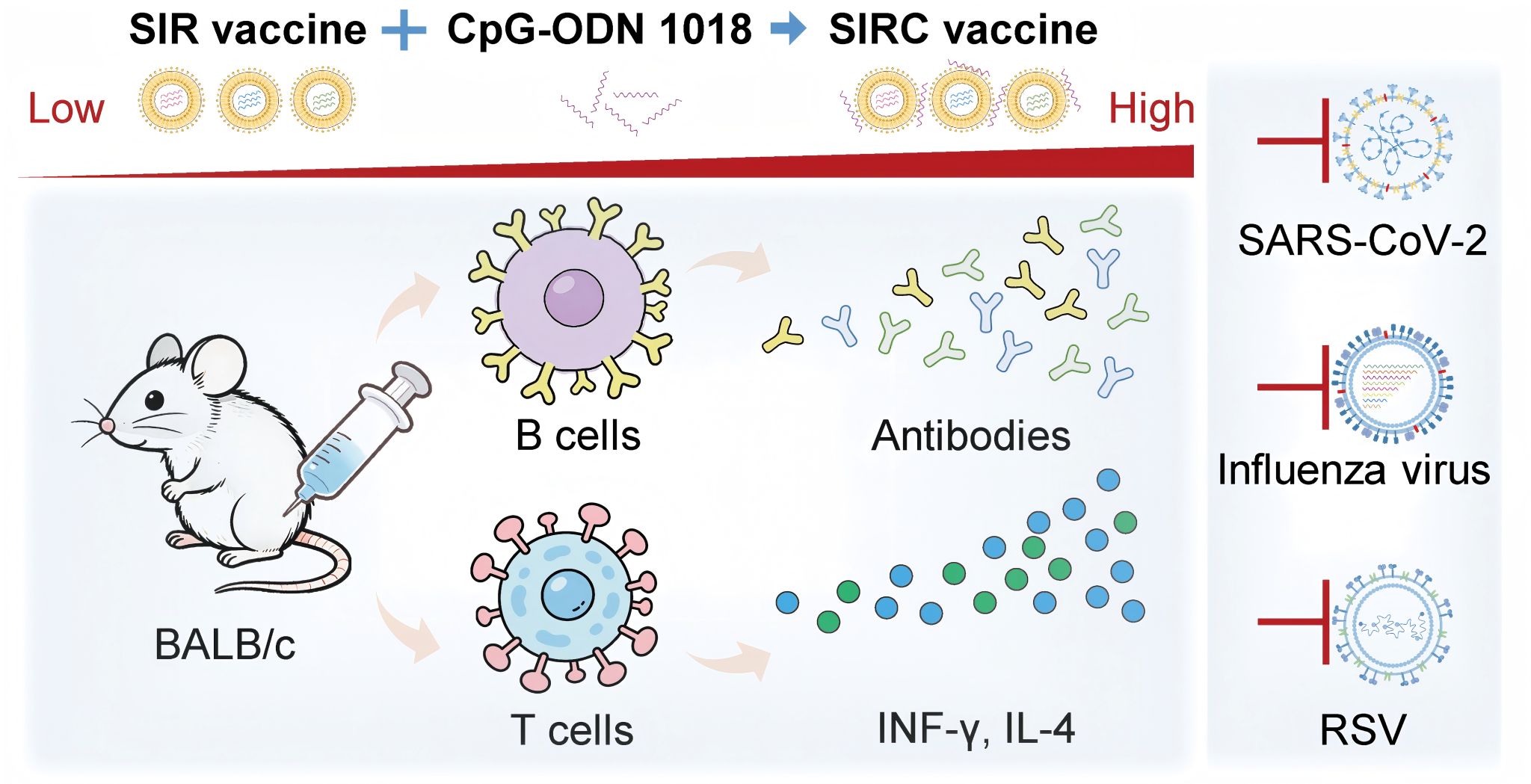
The concurrent circulation of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), influenza virus, and respiratory syncytial virus (RSV) has caused severe coinfections, posing significant public health threats. Developing individual vaccines for these respiratory viruses is hindered by waning immunity and rapid viral mutations. A combination mRNA vaccine streamlines development and administration, reduces healthcare burdens, and offers convenient, compliance-enhancing protection against seasonal illnesses. Here, we present SIR, an 8-valent combination mRNA vaccine designed to simultaneously target SARS-CoV-2, influenza, and RSV. SIR was further optimized into SIRC by incorporating the TLR-9 agonist CpG oligodeoxynucleotides (CpG-ODN) 1018. SIRC induces robust binding and neutralizing antibodies against all three viruses, stimulates Th1-biased antigen-
specific T-cell responses, and generates CD4+ and CD8+ memory T cells. In mice model, SIRC effectively protects against four influenza subtypes (H1N1, H3N2, B/V, and B/Y) and the RSV. Overall, SIRC demonstrates superior antibodies and T cell responses, and provides greater protection in vivo compared to both the individual and SIR vaccines, without causing immune interference. It also maintains a favorable safety and development potential. These findings establish the SIRC as a promising candidate for preventing coinfections involving SARS-CoV-2, influenza, and RSV.
Link: https://pubs.acs.org/doi/10.1021/acsnano.5c14408